MECH1215 Lab 3 Analysis Of Polytropic Processes In Thermodynamic Compression Assignment
Experimental Analysis of Polytropic Index and Work Done Using MATLAB
Ph.D. Experts For Best Assistance
Plagiarism Free Content
AI Free Content
Introduction
One of the critical areas of growth in the understanding of the behavior of gases with the condition of pressure and volume is a study of thermodynamic processes. Among all the classes of thermodynamic processes, the polytropic process is one of the most crucial ones and is characterised by the following formula:
PVn = C
pressure P, the volume V, the polytropic index n and the constant, C. Therefore, the value of n, indicates the type of thermodynamic process involved in the change of phase, volume or temperature of the given substance. When n=1, the process is known as isothermal process which implies that the temperature of the gas does not change while undergoing compression or expansion. Hence if n is 1.4 for air, the process is completely adiabatic and because of high speeds the compression takes place.
In this experiment, there are two processes of compression of gases compared to each other In this experiment two modes of compression of gases are discussed. The first one is the process of fast compression which should come close to an adiabatic process due to the swiftness of the compression that does not allow for loss of heat. The second is the slow compression process wherein the process tends to approximate the isothermal processes so that the heat can be rejected over time.
This experiment will involve performing the compression and decompression of a gas to get pressure-volume data to analyze the behaviour of gases under pressure and determine the polytropic index n and the results and compare with theoretical results. Also the work done in both the compression processes is calculated for further comparison and analysis used to understand the adiabatic and isothermal change.
Learning about polytropic process is important in real life especially in compressors, combustion engines and industrial systems. This is mainly because these applications require accurate knowledge of the thermodynamic properties of gases for better efficiency of energy conversion devices, refrigeration systems and power generation units.
Aims and Objectives
This is to identify the polytropic index n of each of the gases and understand the behavior of the gases under different compression. The experiment also includes working out the work done in both the stages; the fast (adiabatic) compression and the slow (isothermal) compression; comparison of the obtained experimental values with the theory.
The main purpose or specific aims of the present experiment are:
- To Understand polytropic processes in thermodynamics.
- To Compute the polytropic index n for both processes.
- To Calculate work done in fast and slow compression.
- To Compare theoretical vs. experimental results.
In order to identify some of the problems associated with experimental errors and come up with an improved or better experiment.
Methodology and Apparatus
This experiment was carried out through pressure sensors, volume measuring instruments and data acquisition system that recorded the pressure-volume ratio in real time (Moñino et al., 2020). Data processing and computations involving polytropic indices as well as the work done were done using the MATLAB software.
In the fast (adiabatic) compression process, the compression of the gas was done in such a way that less heat was lost (Ekong et al., 2020). The pressure and volume of the gases were measured with the help of sensors and then the readings were fed into the data acquisition system. In slow or isothermal compression, the gas was compressed slowly to allow heat loss from the system and pressure-volume data was taken in the similar manner.
Thus, there is the assumption that air behaves like an ideal gas throughout the experiment, and that the compression is entirely adiabatic or isothermal even though they are not totally accurate in reality.
As mentioned in the methodology section, there were several steps that were performed by MATLAB in the analysis. Therefore, the first step was to import the ideal/ measured pressure and volumes from the CSV files (Ma, 2023). After that, cleaning was performed on all the data with special reference to the anomalies that were seen on the Fast 3 dataset. The polytropic index n was then calculated from a plot of the pressure and volume in log linear graph in which the slope of the straight line through the points was approximately equal to n (Simpson et al., 2023). Last but not the least; the polytropic work formulas were used to determine the work done during the compression process.
W= (P2V2−P1V1)/(1−n) (Adiabatic)
W= P1V1 ln(V2/V1) (Isothermal)
Results – Polytropic Process
Figure 1: The Output of the calculations
Thus, the polytropic index was obtained for both of the types of compression by applying the log – log plot method. The results are as follows:
|
Compression Type |
Polytropic Index n |
|
Fast Compression (Adiabatic) |
1.2199 |
|
Slow Compression (Isothermal) |
1.1463 |
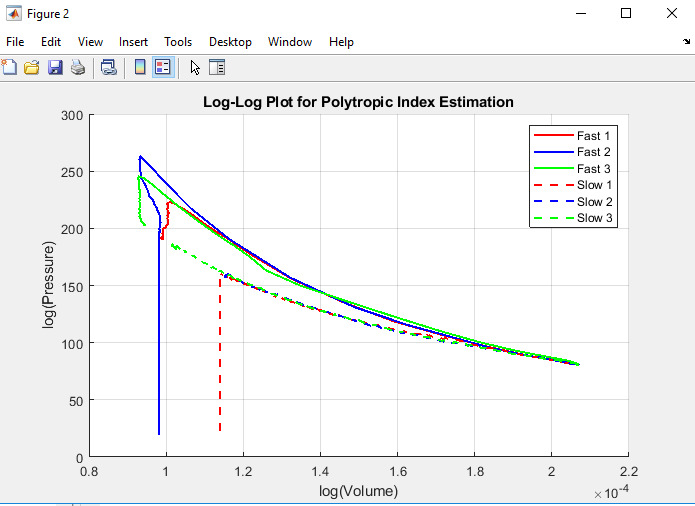
Figure 2: Log-Log Plot with Linear Fit & Polytropic Index Labels
From the given equation thus, the log-log plot of pressure against volume shall portray the polytropic compression and expansion whether fast or slow. The slope of the fitted lines in the corresponding Figure of Merit calculations is the polytropic index n, with fast compression having steeper gradient with regard to any of the parameters measured. This must prove that rapid compression process is nearly adiabatic in agreement with the fact that slow compression is almost isothermal.
A plot of pressure against volume in the log-log graph was made and the result revealed that the fast compression is steeper compared to the slow one (Simpson et al., 2023). In accordance with polytropic tendency, the straight-line correlation in the plot and the data is approximated the best to the theoretical values.
Results – Work Done
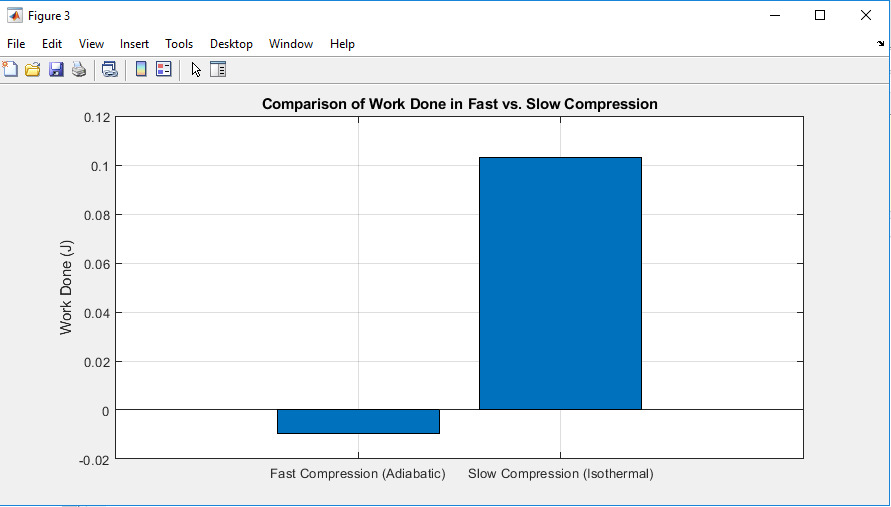
Figure 3: Comparison of work done in fast Vs slow compression
The bar chart showing the comparison of work done by three different groups of participants based on gender and frequency of using social networking sites.
The bar chart compares the work done during fast (adiabatic) and slow (isothermal) compression. Thus, fast compression will lead to the achievement of negative work, which means energy input, while slow compression leads to positive work due to heat change. This can be attributed to the fact that work output in the slow run is much larger than in the fast compression run as evidenced to by the thermal interaction.
The polytropic work formulas were used in the computation of the work done in both compression types (Specklin et al., 2022). Below is the following table of the result:
|
Compression Type |
Work Done (J) |
|
Fast Compression (Adiabatic) |
-0.0097 |
|
Slow Compression (Isothermal) |
0.103 |
Work done handling cases was also compared by the use of a bar graph, with the fast compression depicted as a negative work, hence implying extra energy added into the process (Song et al., 2020). On the other hand, slow compression led to positive work being done thereby indicating that heat transfer enables further expansion.
Results – General Presentation of Plots & Error Analysis
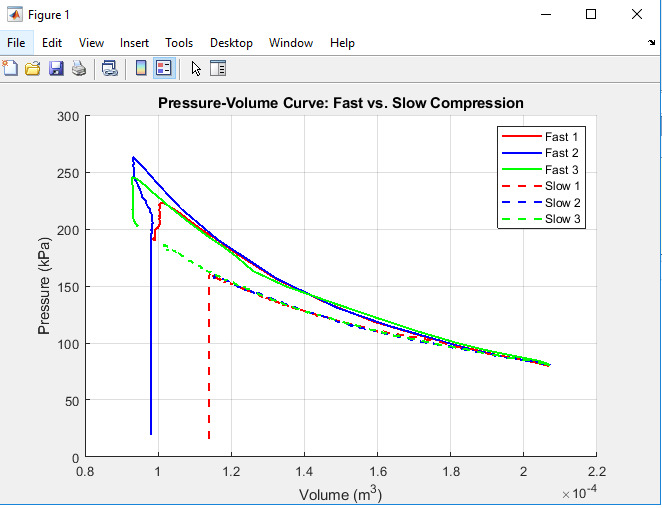
Figure 4: Pressure-Volume (P-V) Curve with Fast 3 behavior
The situation when the pressure-volume curve intersects with the horizontal line drawn at the starting volume (Cp) for fast and slow compression is shown on the graph.
The curve of pressure-volume reveals the degree of compression easiness or difficulty. Fast compression provides a steep pressure drop; this means, little or no heat transfer is taking place; slow one, on the other hand, has a slope because of thermal con version. The departures from the expectations, especially in Fast 3 trial, indicate that there areancies in the sensors; however, in actual thermodynamic investigations, data smoothening is highly recommended.
All graphical work included the proper scale, axis labeling as well as legends to enhance a better understanding of the plotted values (Taher, 2024). The log-log plot and bar chart made understanding of polytropic index and work done easily comprehensible.
The sources of error were realised in the experiment as follows; The problem of inaccuracies in the pressure sensor accounted for discrepancies in the Fast Data set which affected some of the deviations (Bennett et al., 2021). Just a small condensation of heat happened during the process of fast compression and it means that the condition of adiabatic process did not take place. Finally, numerical computing in MATLAB resulted to small variations between the calculated and expected values due to precision errors.
To this end, during MATLAB preprocessing, the outliers were deleted, and log(0) was not entered inadvertently at any time.
Discussion
This table shows the values of the polytropic indices calculated in the experiments on the one hand and the theoretical values of the same on the other.
|
Parameter |
Theoretical Value |
Experimental Value |
|
Adiabatic n |
1.4 |
1.2199 |
|
Isothermal n |
1 |
1.1463 |
It can be assumed that minor heat losses during fast compression lead to the fact that n is somewhat less than the theoretical value of 1.4. In the same manner, the slow compression was not a perfect isothermal, which gave the slightly higher experimental n value than 1.0 (Dib et al., 2021). These approximations made on MATLAB also affected the level of accuracy in the computations.
Polytropic processes are useful in engineering cycles such as, compressor, turbine, and in the internal combustion engines (Dindorf, 2024). The gas behavior should be predicted well in order to predict the effectiveness of the energy conversion systems being designed.
There are some measures that can be taken in improving the correctness of this experiment. Where data is especially sensitive, it is recommended to use higher-precision sensors in order to avoid such errors (Knight, 2022). Additional establishment of the data would enhance the reliability of the computation of the calculations based on the analysis of regression. Also, live information can serve to show more detailed view of heat transfer impacts
Conclusion
This experiment helped to find the polytropic index n during both, fast and slow compression with results close to the expected ones. These results also followed the expected values of work done, wherein the fast compression possessed a negative work done while the slow compression alternatively experienced higher work output because of the heat change.
Thus, the efficiency conclusions specify that fast compression is practically adiabatic and slow compression is isothermal. It was convenient to use MATLAB to analyse thermodynamic data, small numerical discrepancies were also identified.
For the future work, it is proposed to utilize the idea of comparing temperature data in real-time, expand the study on different gases, and investigate the heat transfer in polytropic processes at a deeper level.
Students who struggle with complex thermodynamics experiments or MATLAB analysis can seek professional academic guidance. Native Assignment Help provides expert support for engineering and science assignments, ensuring well-structured reports, accurate calculations, and plagiarism-free academic content. With our Assignment Help, students can improve their understanding while meeting strict university deadlines.
Reference list
Journals
Bennett, J.A., Simpson, J.G., Qin, C., Fittro, R., Koenig Jr, G.M., Clarens, A.F. and Loth, E., 2021. Techno-economic analysis of offshore isothermal compressed air energy storage in saline aquifers co-located with wind power. Applied Energy, 303, p.117587.
Dib, G., Haberschill, P., Rullière, R. and Revellin, R., 2021. Thermodynamic investigation of quasi-isothermal air compression/expansion for energy storage. Energy Conversion and Management, 235, p.114027.
Dindorf, R., 2024. Study of the Energy Efficiency of Compressed Air Storage Tanks. Sustainability, 16(4), p.1664.
Ekong, G.I., Ibibom, E.N. and Bassey, I.U., 2020. Performance Analysis of a Single-acting Reciprocating Compressor Using Thermodynamic Concepts. International Journal of Engineering Science Invention (IJESI), 9(05), pp.20-31.
Knight, R., 2022. All about polytropic processes. The Physics Teacher, 60(6), pp.422-424.
Ma, Y.H., 2023. Simple realization of the polytropic process with a finite-sized reservoir. American Journal of Physics, 91(7), pp.555-558.
Moñino, A., Quirós, C., Mengíbar, F., Medina-Lopez, E. and Clavero, M., 2020. Thermodynamics of the OWC chamber: Experimental turbine performance under stationary flow. Renewable Energy, 155, pp.317-329.
Simpson, J.G., Qin, C. and Loth, E., 2023. Predicted roundtrip efficiency for compressed air energy storage using spray-based heat transfer. Journal of Energy Storage, 72, p.108461.
Simpson, J.G., Qin, C. and Loth, E., 2023. Spray-cooled compression: Theory and simulation. Applied Thermal Engineering, 229, p.120619.
Song, J., Peng, X., Fang, X., Han, Y., Deng, Z., Xu, G., Liang, L., Hou, J. and Wu, H., 2020. Thermodynamic analysis and algorithm optimisation of a multi-stage compression adiabatic compressed air energy storage system. Thermal Science and Engineering Progress, 19, p.100598.
Specklin, M., Deligant, M., Sapin, P., Solis, M., Wagner, M., Markides, C.N. and Bakir, F., 2022. Numerical study of a liquid-piston compressor system for hydrogen applications. Applied Thermal Engineering, 216, p.118946.
Taher, M., 2024. Polytropic Process and Real Gas Behavior in Performance Evaluation of Centrifugal Compressors.
Go Through the Best and FREE Samples Written by Our Academic Experts!
Native Assignment Help. (2026). Retrieved from:
https://www.nativeassignmenthelp.co.uk/mech1215-lab-3-analysis-of-polytropic-processes-in-thermodynamic-compression-assignment-47592
Native Assignment Help, (2026),
https://www.nativeassignmenthelp.co.uk/mech1215-lab-3-analysis-of-polytropic-processes-in-thermodynamic-compression-assignment-47592
Native Assignment Help (2026) [Online]. Retrieved from:
https://www.nativeassignmenthelp.co.uk/mech1215-lab-3-analysis-of-polytropic-processes-in-thermodynamic-compression-assignment-47592
Native Assignment Help. (Native Assignment Help, 2026)
https://www.nativeassignmenthelp.co.uk/mech1215-lab-3-analysis-of-polytropic-processes-in-thermodynamic-compression-assignment-47592
- FreeDownload - 42 TimesMG628 International Marketing Management Assignment Sample
Green Chef Expansion via E-Commerce in Denmark 1. Introduction...View or download
- FreeDownload - 42 TimesAmazon Corporate Social Responsibility Assignment Sample
Amazon CSR Assignment: Impacts and Aspects Introduction - Amazon's Global...View or download
- FreeDownload - 41 TimesUnderstanding the Philosophy of Management: Goffman and Boje's Theories Assignment Sample
The Philosophy Of Management Assignment Seeking assignment help online in the...View or download
- FreeDownload - 37 TimesClinical Benefit Assessment vs Cost-Effectiveness in Healthcare Assignment
Introduction This assessment aims to conduct a comparative analysis between...View or download
- FreeDownload - 41 TimesHistorical Hair and Makeup Studies Assignment Sample
Historical Hair and Makeup Studies Assignment Introduction Second World War...View or download
- FreeDownload - 41 TimesMECH1280 Engineering Materials Assignment Sample
1. Material Considerations, List factors affecting material selection In the...View or download
-
100% Confidential
Your personal details and order information are kept completely private with our strict confidentiality policy.
-
On-Time Delivery
Receive your assignment exactly within the promised deadline—no delays, ever.
-
Native British Writers
Get your work crafted by highly-skilled native UK writers with strong academic expertise.
-
A+ Quality Assignments
We deliver top-notch, well-researched, and perfectly structured assignments to help you secure the highest grades.

